The article is published as part of an exclusive content-sharing agreement with neo-trans.blog.
Following government approval of a new therapy to treat a rare genetic skin disorder, Abeona Therapeutics Inc. revealed through public records that it plans another expansion of its footprint next to its Midtown Cleveland headquarters.
The 10-year-old, publicly traded (Nasdaq: ABEO) therapeutics venture, based in Building Three of the Midtown Tech Park, 6555 Carnegie Ave., revealed that it plans to invest $2.85 million to expand into a neighboring building of the Midtown Tech Park. It is not known how many new jobs will come with this expansion, but is likely fewer than two dozen.
In a permit application submitted last week to the Cleveland Building Department, the company plans to fit-out a 7,587-square-foot space with laboratory facilities on the second-floor of Midtown Tech Park’s Building One, 6700 Euclid Ave.
This is the latest major expansion by Abeona at the Midtown Tech Park. Its last was five years ago when the company leased and modified 12,000 square feet of space on the northwest corner of the second floors of Building One. The planned expansion is on the southwest corner of the same building.
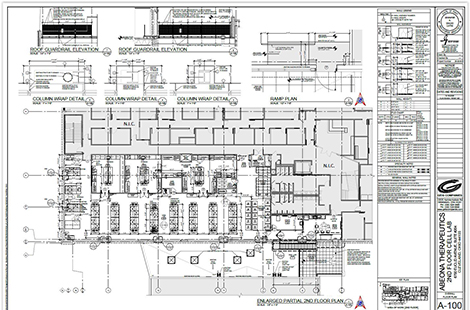
Building Two at the tech park is at 7000 Euclid Ave. The 240,000-square-foot complex of office, medical, and laboratory space was developed over the past 15 years by Hemingway Development, then an affiliate of Geis Companies. Midtown Tech Park continues to be owned by an affiliate of Geis.
Abeona received in April U.S. Food and Drug Administration (FDA) approval of ZEVASKYN™ (prademagene zamikeracel). ZEVASKYN is the first and only autologous cell-based gene therapy for the treatment of wounds in adults and pediatric patients with recessive dystrophic epidermolysis bullosa (RDEB).
RDEB is a genetic disorder that causes extremely fragile skin, leading to blisters and open sores from minor injuries or friction. After FDA approval, Abeona announced it had entered into a definitive asset purchase agreement to sell its Rare Pediatric Disease Priority Review Voucher (PRV) for gross proceeds of $155 million upon the closing of the transaction.
The company has a fully integrated cell and gene therapy Current Good Manufacturing Practices facility in Building Three at Midtown Tech Park. It serves as the manufacturing site for ZEVASKYN commercial production. But with the growth of that activity, additional space was needed for cell and gene therapies for other serious diseases.

“With proceeds from this PRV sale, we have sufficient cash for more than two years of operating expenses without the need for capital infusion and not accounting for ZEVASKYN sales,” said Joe Vazzano, Chief Financial Officer of Abeona in a written statement. “Furthermore, with ZEVASKYN becoming available to treat patients beginning third quarter of 2025, we anticipate becoming profitable in early 2026.”
Abeona, a commercial-stage biopharmaceutical company developing cell and gene therapies for serious diseases, was founded in 1974 as Access Pharmaceuticals, Inc. In 2015, it transformed into Abeona Therapeutics to focus on gene and cell therapies.
The company is developing additional treatments, including adeno-associated virus (AAV)-based gene therapies for ophthalmic diseases with high unmet medical need.
There are nearly 200 health-technology and high-technology employers in Cleveland’s Health Tech Corridor which runs along and between Cedar and Chester avenues, from University Circle to the Campus District near Downtown Cleveland. Fifty of those firms are in Midtown proper, according to MidTown Cleveland.
For more updates about Cleveland, sign up for our Cleveland Magazine Daily newsletter, delivered to your inbox six times a week.
Cleveland Magazine is also available in print, publishing 12 times a year with immersive features, helpful guides and beautiful photography and design.




